


Next: Computer Implementation of
Up: Image Analysis
Previous: Comparisons and Counterexamples
Alfred S. Carasso, ACMD
Margaret Daube-Witherspoon, National Institutes of Health
Developed over 20 years ago, Positron Emission Tomography, (PET), is currently
the most widely used technology for studying physiological and
anatomical abnormalities in the brain associated with such disorders as
schizophrenia, dementia, Alzheimer's disease, mood disorders, and alcoholism.
A positron emitting radionuclide is used to tag glucose molecules in their
course through the brain. The metabolic rate of glucose measures cerebral
function, and indicates the extent to which various regions of the brain
are working, or failing to work, in response to a given task. Emitted positrons
travel approximately 1-2 mm before colliding with an electron in an
annihilating reaction that produces gamma ray photons. A gamma detector
records every such positron emission, eventually leading to an image of the
distribution of the glucose tracer. However, owing to scattering of the emitted
positrons prior to annihilation, and to detector effects, the resulting PET
images are blurred and sometimes very noisy.
An ongoing research project between ACMD and the Nuclear Medicine Branch at the
National Institutes of Health, seeks to improve the quality
of these images through Image Restoration.
Empirical studies at NIH indicate that the blur can be described by a Gaussian
point spread function with a full width at half maximum equal to 7.1 mm. The
SECB method developed by Carasso is exceptionally well-suited for this class
of problems for the following reasons. Unlike many deblurring algorithms, the
SECB method does not impose a smoothness constraint on the unknown true
image. This is an important consideration in medical imaging where there may
be genuine singularities or hot spots in the true image, associated with
lesions or tumors. A deconvolution scheme that enforces smoothness may fail to
detect such singularities. The SECB method uses three parameters that express
the regularizing a-priori information. Two of these parameters can be set at
conservative values at the outset, leaving one parameter, K, typically lying
between 0 and 5, to be adjusted interactively. For the 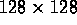 images produced by the NIH PET scanner, the direct SECB method can perform
20 trial restorations, each with a different value of K, in 20 seconds on
an SGI Indigo. By displaying these results and readjusting K as necessary,
the optimal value of that parameter can be quickly located. In contrast,
with iterative deblurring algorithms where several thousand iterations may
be needed to converge to a single trial restoration, such necessary interactive
adjustment becomes a prohibitive task, often requiring week-long computations.
images produced by the NIH PET scanner, the direct SECB method can perform
20 trial restorations, each with a different value of K, in 20 seconds on
an SGI Indigo. By displaying these results and readjusting K as necessary,
the optimal value of that parameter can be quickly located. In contrast,
with iterative deblurring algorithms where several thousand iterations may
be needed to converge to a single trial restoration, such necessary interactive
adjustment becomes a prohibitive task, often requiring week-long computations.
Several hundred such PET images have been deblurred to date, with the results
transmitted back to NIH for comprehensive statistical studies. In most cases,
SECB deblurring reveals interesting
structures and centers of activity that are not evident in the blurred image.
In some cases, excessive amounts of correlated noise in the
blurred image produce visible noise artifacts in the deblurred image, causing
difficulty in interpreting the results. Future plans call for studying a
different class of PET images that exhibit substantial blur but relatively
little noise. In addition, controlled deblurring experiments are planned with
PET images of artificial plastic brain phantoms. Here, the unblurred
image is known, and can be used to verify the results of the SECB technique.



Next: Computer Implementation of
Up: Image Analysis
Previous: Comparisons and Counterexamples
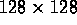 images produced by the NIH PET scanner, the direct SECB method can perform
20 trial restorations, each with a different value of K, in 20 seconds on
an SGI Indigo. By displaying these results and readjusting K as necessary,
the optimal value of that parameter can be quickly located. In contrast,
with iterative deblurring algorithms where several thousand iterations may
be needed to converge to a single trial restoration, such necessary interactive
adjustment becomes a prohibitive task, often requiring week-long computations.
images produced by the NIH PET scanner, the direct SECB method can perform
20 trial restorations, each with a different value of K, in 20 seconds on
an SGI Indigo. By displaying these results and readjusting K as necessary,
the optimal value of that parameter can be quickly located. In contrast,
with iterative deblurring algorithms where several thousand iterations may
be needed to converge to a single trial restoration, such necessary interactive
adjustment becomes a prohibitive task, often requiring week-long computations.