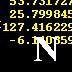 Return to periodic table
Return to periodic tableThe ground electronic configurations of the elements H through U and their first cations are given below. The notation [X] indicates that all subshells associated with the noble gas X are fully occupied.
Electronic Configuration
Z Symbol Neutral Positive ion
1 H 1s^1 -
2 He 1s^2 1s^1
3 Li [He] 2s^1 1s^2
4 Be [He] 2s^2 [He] 2s^1
5 B [He] 2s^2 2p^1 [He] 2s^2
6 C [He] 2s^2 2p^2 [He] 2s^2 2p^1
7 N [He] 2s^2 2p^3 [He] 2s^2 2p^2
8 O [He] 2s^2 2p^4 [He] 2s^2 2p^3
9 F [He] 2s^2 2p^5 [He] 2s^2 2p^4
10 Ne [He] 2s^2 2p^6 [He] 2s^2 2p^5
11 Na [Ne] 3s^1 [He] 2s^2 2p^6
12 Mg [Ne] 3s^2 [Ne] 3s^1
13 Al [Ne] 3s^2 3p^1 [Ne] 3s^2
14 Si [Ne] 3s^2 3p^2 [Ne] 3s^2 3p^1
15 P [Ne] 3s^2 3p^3 [Ne] 3s^2 3p^2
16 S [Ne] 3s^2 3p^4 [Ne] 3s^2 3p^3
17 Cl [Ne] 3s^2 3p^5 [Ne] 3s^2 3p^4
18 Ar [Ne] 3s^2 3p^6 [Ne] 3s^2 3p^5
19 K [Ar] 4s^1 [Ne] 3s^2 3p^6
20 Ca [Ar] 4s^2 [Ar] 4s^1
21 Sc [Ar] 3d^1 4s^2 [Ar] 3d^1 4s^1
22 Ti [Ar] 3d^2 4s^2 [Ar] 3d^2 4s^1
23 V [Ar] 3d^3 4s^2 [Ar] 3d^4
24 Cr [Ar] 3d^5 4s^1 [Ar] 3d^5
25 Mn [Ar] 3d^5 4s^2 [Ar] 3d^5 4s^1
26 Fe [Ar] 3d^6 4s^2 [Ar] 3d^6 4s^1
27 Co [Ar] 3d^7 4s^2 [Ar] 3d^8
28 Ni [Ar] 3d^8 4s^2 [Ar] 3d^9
29 Cu [Ar] 3d^10 4s^1 [Ar] 3d^10
30 Zn [Ar] 3d^10 4s^2 [Ar] 3d^10 4s^1
31 Ga [Ar] 3d^10 4s^2 4p^1 [Ar] 3d^10 4s^2
32 Ge [Ar] 3d^10 4s^2 4p^2 [Ar] 3d^10 4s^2 4p^1
33 As [Ar] 3d^10 4s^2 4p^3 [Ar] 3d^10 4s^2 4p^2
34 Se [Ar] 3d^10 4s^2 4p^4 [Ar] 3d^10 4s^2 4p^3
35 Br [Ar] 3d^10 4s^2 4p^5 [Ar] 3d^10 4s^2 4p^4
36 Kr [Ar] 3d^10 4s^2 4p^6 [Ar] 3d^10 4s^2 4p^5
37 Rb [Kr] 5s^1 [Ar] 3d^10 4s^2 4p^6
38 Sr [Kr] 5s^2 [Kr] 5s^1
39 Y [Kr] 4d^1 5s^2 [Kr] 5s^2
40 Zr [Kr] 4d^2 5s^2 [Kr] 4d^2 5s^1
41 Nb [Kr] 4d^4 5s^1 [Kr] 4d^4
42 Mo [Kr] 4d^5 5s^1 [Kr] 4d^5
43 Tc [Kr] 4d^5 5s^2 [Kr] 4d^5 5s^1
44 Ru [Kr] 4d^7 5s^1 [Kr] 4d^7
45 Rh [Kr] 4d^8 5s^1 [Kr] 4d^8
46 Pd [Kr] 4d^10 [Kr] 4d^9
47 Ag [Kr] 4d^10 5s^1 [Kr] 4d^10
48 Cd [Kr] 4d^10 5s^2 [Kr] 4d^10 5s^1
49 In [Kr] 4d^10 5s^2 5p^1 [Kr] 4d^10 5s^2
50 Sn [Kr] 4d^10 5s^2 5p^2 [Kr] 4d^10 5s^2 5p^1
51 Sb [Kr] 4d^10 5s^2 5p^3 [Kr] 4d^10 5s^2 5p^2
52 Te [Kr] 4d^10 5s^2 5p^4 [Kr] 4d^10 5s^2 5p^3
53 I [Kr] 4d^10 5s^2 5p^5 [Kr] 4d^10 5s^2 5p^4
54 Xe [Kr] 4d^10 5s^2 5p^6 [Kr] 4d^10 5s^2 5p^5
55 Cs [Xe] 6s^1 [Kr] 4d^10 5s^2 5p^6
56 Ba [Xe] 6s^2 [Xe] 6s^1
57 La [Xe] 5d^1 6s^2 [Xe] 5d^2
58 Ce [Xe] 4f^1 5d^1 6s^2 [Xe] 4f^1 5d^2
59 Pr [Xe] 4f^3 6s^2 [Xe] 4f^3 6s^1
60 Nd [Xe] 4f^4 6s^2 [Xe] 4f^4 6s^1
61 Pm [Xe] 4f^5 6s^2 [Xe] 4f^5 6s^1
62 Sm [Xe] 4f^6 6s^2 [Xe] 4f^6 6s^1
63 Eu [Xe] 4f^7 6s^2 [Xe] 4f^7 6s^1
64 Gd [Xe] 4f^7 5d^1 6s^2 [Xe] 4f^7 5d^1 6s^1
65 Tb [Xe] 4f^9 6s^2 [Xe] 4f^9 6s^1
66 Dy [Xe] 4f^10 6s^2 [Xe] 4f^10 6s^1
67 Ho [Xe] 4f^11 6s^2 [Xe] 4f^11 6s^1
68 Er [Xe] 4f^12 6s^2 [Xe] 4f^12 6s^1
69 Tm [Xe] 4f^13 6s^2 [Xe] 4f^13 6s^1
70 Yb [Xe] 4f^14 6s^2 [Xe] 4f^14 6s^1
71 Lu [Xe] 4f^14 5d^1 6s^2 [Xe] 4f^14 6s^2
72 Hf [Xe] 4f^14 5d^2 6s^2 [Xe] 4f^14 5d^1 6s^2
73 Ta [Xe] 4f^14 5d^3 6s^2 [Xe] 4f^14 5d^3 6s^1
74 W [Xe] 4f^14 5d^4 6s^2 [Xe] 4f^14 5d^4 6s^1
75 Re [Xe] 4f^14 5d^5 6s^2 [Xe] 4f^14 5d^5 6s^1
76 Os [Xe] 4f^14 5d^6 6s^2 [Xe] 4f^14 5d^6 6s^1
77 Ir [Xe] 4f^14 5d^7 6s^2 [Xe] 4f^14 5d^7 6s^1
78 Pt [Xe] 4f^14 5d^9 6s^1 [Xe] 4f^14 5d^9
79 Au [Xe] 4f^14 5d^10 6s^1 [Xe] 4f^14 5d^10
80 Hg [Xe] 4f^14 5d^10 6s^2 [Xe] 4f^14 5d^10 6s^1
81 Tl [Xe] 4f^14 5d^10 6s^2 6p^1 [Xe] 4f^14 5d^10 6s^2
82 Pb [Xe] 4f^14 5d^10 6s^2 6p^2 [Xe] 4f^14 5d^10 6s^2 6p^1
83 Bi [Xe] 4f^14 5d^10 6s^2 6p^3 [Xe] 4f^14 5d^10 6s^2 6p^2
84 Po [Xe] 4f^14 5d^10 6s^2 6p^4 [Xe] 4f^14 5d^10 6s^2 6p^3
85 At [Xe] 4f^14 5d^10 6s^2 6p^5 [Xe] 4f^14 5d^10 6s^2 6p^4
86 Rn [Xe] 4f^14 5d^10 6s^2 6p^6 [Xe] 4f^14 5d^10 6s^2 6p^5
87 Fr [Rn] 7s^1 [Xe] 4f^14 5d^10 6s^2 6p^6
88 Ra [Rn] 7s^2 [Rn] 7s^1
89 Ac [Rn] 6d^1 7s^2 [Rn] 7s^2
90 Th [Rn] 6d^2 7s^2 [Rn] 6d^2 7s^1
91 Pa [Rn] 5f^2 6d^1 7s^2 [Rn] 5f^2 7s^2
92 U [Rn] 5f^3 6d^1 7s^2 [Rn] 5f^3 7s^2
Sources:
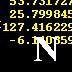 Return to periodic table
Return to periodic table